Cell Cycle Control and Chromatin DNA Labelling
MATERIALS
METHODS
1. Culture the cells using cover slip-bottomed petridish in room B134. For imaging, the cell density is about 70%.
2. Bring the dishes to room B220 (or somewhere else you think it’s proper). Briefly wash the cell with 1x phosphate-buffered saline (PBS) (pH 7.4) two times. Just shake the dish by hand gently. Add or remove PBS by tips carefully.
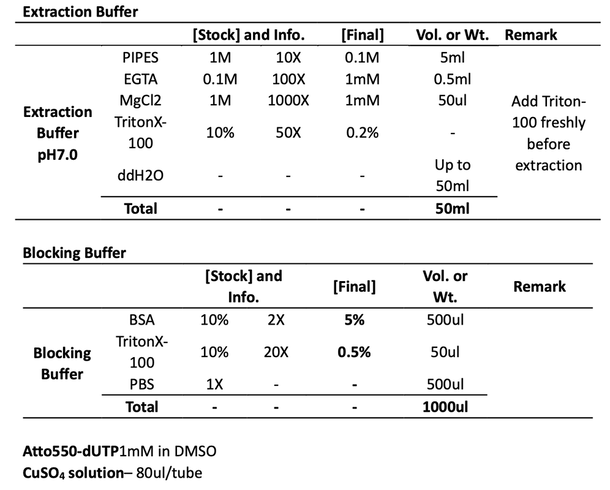
3. Extraction for 1 min with 0.2% Triton X-100 in a pH 7 buffer consisting of 0.1 M PIPES, 1 mM ethylene glycol tetraacetic acid, and 1 mM magnesium chloride. Shaking (<1Hz).
PS: Triton X-100 is added freshly just before extraction. For one dish, we need ~1ml extraction buffer at room temperature.[BP1]
4. Fixation in 4% PFA and 0.1% GA in PBS, aside for 10min.
PS: Fixation buffer is made freshly. For one dish, we need ~500ul fixation buffer at room temperature.
5. Rinse with PBS. Reduction with ~10mM NaBH4 in PBS for 5min. Shaking (<1Hz).
PS: NaBH4 is made freshly, and it will produce lots of bubbles. For one dish, we need ~1ml fixation buffer at room temperature.
6. Briefly wash with 1xPBS twice.
7. Block and permeabilization with 3% BSA and 0.5% Triton X-100 in PBS for 30 min, no shaking.
PS: Triton X-100 is added freshly just before use. For one dish, we need 500-1000ul blocking buffer at room temperature.
8. Stain without shaking for 40 min with the specific primary antibody. For one dish, we need 200-300ul, make sure it covers your cell.
9. Wash with 1x PBS 3 times, 5min/time, shaking (<1Hz).
10. Stain without shaking for 60 min with labeled second antibody (diluted to 2.5ug/ml inblocking buffer. For one dish, we need ~200ul).
11. Washing with 1x PBS 3 times, 5min/time, shaking (<1Hz).
12. Post-fixation in a mixture of 4% PFA and 0.1% GA in PBS for 10 min.
PS: Fixation buffer is made freshly. For one dish, we need ~500ul fixation buffer at room temperature. No shaking.
13. Briefly wash the cell with 1x phosphate-buffered saline (PBS) two times. Just shake the dish by hand gently. Add or remove PBS by tips.
14. Do the imaging (use imaging buffer to replace PBS just before imaging).
[BP1]Extraction is ONLY for MT imaging
1. Culture the cells using cover slip-bottomed petridish in room B134. For imaging, the cell density is about 70%.
2. Bring the dishes to room B220 (or somewhere else you think it’s proper). Briefly wash the cell with 1x phosphate-buffered saline (PBS) (pH 7.4) two times. Just shake the dish by hand gently. Add or remove PBS by tips carefully.
3. Extraction for 1 min with 0.2% Triton X-100 in a pH 7 buffer consisting of 0.1 M PIPES, 1 mM ethylene glycol tetraacetic acid, and 1 mM magnesium chloride. Shaking (<1Hz).
PS: Triton X-100 is added freshly just before extraction. For one dish, we need ~1ml extraction buffer at room temperature.[BP1]
4. Fixation in 4% PFA and 0.1% GA in PBS, aside for 10min.
PS: Fixation buffer is made freshly. For one dish, we need ~500ul fixation buffer at room temperature.
5. Rinse with PBS. Reduction with ~10mM NaBH4 in PBS for 5min. Shaking (<1Hz).
PS: NaBH4 is made freshly, and it will produce lots of bubbles. For one dish, we need ~1ml fixation buffer at room temperature.
6. Briefly wash with 1xPBS twice.
7. Block and permeabilization with 3% BSA and 0.5% Triton X-100 in PBS for 30 min, no shaking.
PS: Triton X-100 is added freshly just before use. For one dish, we need 500-1000ul blocking buffer at room temperature.
8. Stain without shaking for 40 min with the specific primary antibody. For one dish, we need 200-300ul, make sure it covers your cell.
9. Wash with 1x PBS 3 times, 5min/time, shaking (<1Hz).
10. Stain without shaking for 60 min with labeled second antibody (diluted to 2.5ug/ml inblocking buffer. For one dish, we need ~200ul).
11. Washing with 1x PBS 3 times, 5min/time, shaking (<1Hz).
12. Post-fixation in a mixture of 4% PFA and 0.1% GA in PBS for 10 min.
PS: Fixation buffer is made freshly. For one dish, we need ~500ul fixation buffer at room temperature. No shaking.
13. Briefly wash the cell with 1x phosphate-buffered saline (PBS) two times. Just shake the dish by hand gently. Add or remove PBS by tips.
14. Do the imaging (use imaging buffer to replace PBS just before imaging).
[BP1]Extraction is ONLY for MT imaging